Chelated Iron for Modern Greenhouse Fertigation: Why Stable Iron Chelation Matters

Iron Nutrition in Global Greenhouse Production
Iron plays a critical role in modern crop production. Although required in relatively small quantities compared with macronutrients such as nitrogen or potassium, iron is indispensable for plant metabolism and healthy growth. Without an adequate supply of iron, crops quickly show symptoms of deficiency, most notably the yellowing of young leaves known as iron chlorosis.
In modern horticulture, especially in large commercial greenhouse operations, maintaining a consistent supply of micronutrients is essential. Many greenhouse complexes now operate highly controlled fertigation systems that deliver precise nutrient solutions directly to plant roots through irrigation networks. These systems often recycle water and rely on technologies such as ultraviolet sterilisation to maintain hygiene and prevent the spread of plant pathogens.
Within these advanced production environments, the form in which nutrients are delivered becomes extremely important. Iron must remain soluble, stable in solution and biologically available as it moves through irrigation lines and root zones.
S-Chelate Plant Nutrition has developed advanced chelation technology designed to meet these demands. S-Chelate-O Iron is used in micronutrient formulations supplied to greenhouse growers worldwide. S-Chelate Plant Nutrition has developed advanced chelation technology designed to meet these demands. S-Chelate-O Iron is formulated to deliver stable, soluble iron for modern fertigation systems used in greenhouse production worldwide.
Key Takeaways
- Iron is an essential micronutrient required for chlorophyll formation, photosynthesis and healthy plant development.
- Chelated iron keeps iron soluble and biologically available for plant uptake in fertigation systems.
- Traditional fertilisers such as ferrous sulphate may struggle with solubility and stability in irrigation water.
- Some conventional iron chelates can interfere with UV water sterilisation in recirculating irrigation systems.
- S-Chelate-O Iron is designed to remain stable, soluble and compatible with modern greenhouse irrigation technology.
Did you know? Some iron fertilisers can reduce the effectiveness of UV water sterilisation systems in greenhouses because of their strong colour.
Why Iron Is Essential for Plant Health

Iron is one of the most important micronutrients involved in plant physiology. Even though plants require iron in small concentrations, its role within plant cells is fundamental for a wide range of biochemical processes.
Key Functions of Iron in Plants
Iron contributes to several critical plant processes, including:
- Chlorophyll formation
- Photosynthesis
- Enzyme activation
- Energy transfer within plant cells
- Nitrogen metabolism
Iron is not a structural component of chlorophyll itself, but it is essential for the biochemical reactions that produce chlorophyll within plant tissues. Without adequate iron availability, plants cannot maintain healthy green foliage.
Iron Chlorosis
One of the most recognisable symptoms of iron deficiency is iron chlorosis. This condition occurs when plants cannot access sufficient iron for chlorophyll production.
Typical symptoms include:
- Yellowing of young leaves
- Green veins contrasting against yellow tissue
- Reduced plant vigour
- Lower crop yields
Greenhouse crops such as tomatoes, cucumbers, peppers and ornamentals are particularly sensitive to iron deficiencies. Even short periods of limited iron availability can affect plant growth and crop quality.
Why Availability Matters
The total iron concentration in irrigation water or fertiliser solutions does not necessarily reflect how much iron plants can actually absorb. Iron can easily react with other elements and form insoluble compounds.
Factors influencing iron availability include:
- Irrigation water pH
- Nutrient solution chemistry
- Interactions with other fertilisers
- Stability of the iron source used
For this reason, professional horticulture increasingly relies on chelated micronutrients to ensure reliable nutrient uptake.
Why Chelated Iron Is the Most Effective Way to Supply Iron

Chelated iron has become the preferred method for supplying iron to crops in modern horticulture systems. Chelation refers to a chemical structure that surrounds and protects the iron ion, allowing it to remain stable in solution and available for plant uptake.
Without this protection, iron ions readily react with oxygen, phosphates and other compounds present in irrigation water. These reactions quickly produce insoluble particles that plants cannot absorb.
How Chelation Works
A chelating molecule binds tightly to the iron ion and forms a stable complex. This complex keeps the iron dissolved in the nutrient solution and maintains solubility in alkaline environments. Akali reacts with other forms of Iron to make it fall out of solution and become biologically unavailable.
This stability allows the iron to remain available for plant roots, even as it travels through irrigation systems.
Advantages of Chelated Iron
Chelated iron offers several benefits for modern agriculture:
- Improved solubility in nutrient solutions
- Greater stability across different pH conditions
- Reduced precipitation in irrigation systems
- More reliable plant uptake
- Compatibility with automated fertigation systems
These advantages are particularly important in high technology greenhouse environments where precise nutrient delivery is essential.
Chelation and Modern Greenhouse Production
Large greenhouse operations rely on fertigation systems that distribute nutrient solutions through extensive irrigation networks. Any fertiliser used in these systems must dissolve easily and remain stable during transport.
Chelated micronutrients provide this reliability, ensuring that crops receive consistent nutrition throughout the production cycle.
The Limitations of Basic Iron Fertilisers
Traditional iron fertilisers were developed long before modern greenhouse fertigation systems became common. While these products can still provide iron under certain conditions, they often perform poorly in advanced irrigation systems.
Ferrous Sulphate
Ferrous sulphate is often used because it is inexpensive. However, it has limited solubility and the iron can quickly become unavailable to plants. In fertigation systems this can lead to inconsistent iron availability and reduced nutrient efficiency.
- Limited solubility in water
- Rapid oxidation when exposed to air
- Formation of insoluble compounds in alkaline conditions
These properties mean that iron supplied as ferrous sulphate can quickly become unavailable to plants.
Practical Challenges in Irrigation Systems
In modern greenhouse fertigation systems, poorly soluble fertilisers can create several operational problems.
Potential issues include:
- Precipitation within nutrient tanks
- Sediment accumulation in irrigation pipes
- Blocked emitters and drippers
- Inconsistent nutrient delivery to crops
Because of these challenges, greenhouse growers typically rely on more advanced micronutrient formulations that remain fully soluble and stable in irrigation water.
Challenges with Traditional Iron Chelates
Chelation greatly improves iron availability, but not all chelated iron products behave the same way in modern greenhouse systems.
Different chelating agents have different chemical properties. These properties influence how the iron behaves under different conditions such as pH levels or exposure to ultraviolet light.
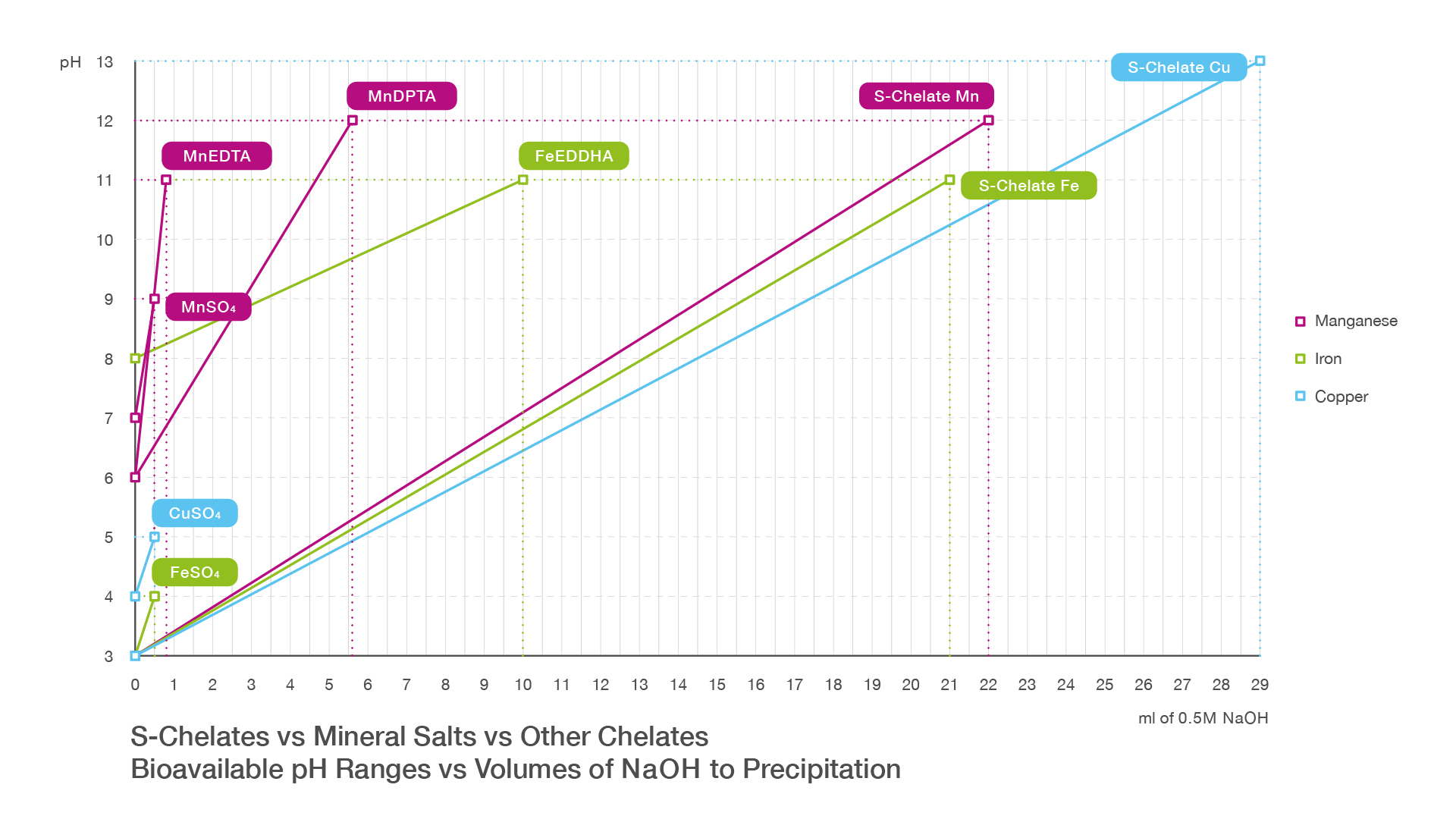
EDDHA Chelated Iron
EDDHA is a commonly used iron chelate, particularly in alkaline growing conditions. However, EDDHA fertilisers typically produce a deep purple solution.
This strong colour absorbs ultraviolet light. In irrigation systems that rely on UV sterilisation to disinfect recirculated water, this can reduce the effectiveness of the treatment process.
For greenhouse operations using UV water sterilisation, the transparency of the nutrient solution becomes an important consideration.
EDTA and DTPA Chelates
Other common iron chelates include EDTA and DTPA.
These products perform well under certain conditions but their stability can decrease as pH levels rise. In irrigation systems with alkaline water, this can reduce the effectiveness of the iron source.
S-Chelate Plant Nutrition has developed S-Chelate technology specifically to provide reliable micronutrient availability across a wider range of pH conditions.
S-Chelate-O Iron Technology for Stable Iron Nutrition
S-Chelate-O Iron is part of the micronutrient range developed by S-Chelate Plant Nutrition. The technology focuses on maintaining metal ions in a stable and biologically available form for plant uptake.
Key Product Characteristics
S-Chelate-O Iron contains:
- 13 percent elemental iron
- Water soluble powder formulation
- Chelated iron designed for high biological availability
The product has been developed to remain stable across a wide pH range, allowing it to perform effectively in different irrigation environments.
Physical Properties
S-Chelate-O Iron is supplied as a water soluble powder with the following characteristics:
| Property | Specification |
| Iron content | 13 percent |
| Product form | Water soluble powder |
| Appearance | Brown to pale green |
| Stability range | Approx. pH 3 to pH 11 |
When dissolved, S-Chelate-O Iron produces a light green solution that remains transparent enough for UV water treatment systems.
Benefits for Fertigation Systems
S-Chelate-O Iron is designed to support modern horticultural production systems.
Key advantages include:
- High solubility for rapid mixing
- Stable nutrient solutions
- Reliable micronutrient availability
- Compatibility with modern irrigation technologies
Why UV Compatibility Matters in Modern Greenhouse Irrigation

Water hygiene is a major priority in modern greenhouse production. Many large greenhouse operations recycle irrigation water to reduce waste and improve sustainability.
Recirculated water must be disinfected to prevent the spread of plant pathogens.
UV Water Treatment
Ultraviolet sterilisation is widely used to disinfect irrigation water. UV systems expose water to ultraviolet light, which damages the DNA of microorganisms and prevents them from spreading disease.
For UV sterilisation to work effectively, light must be able to pass through the irrigation solution.
The Importance of Nutrient Transparency
If fertilisers strongly absorb or block UV light, they may reduce the efficiency of the sterilisation system.
Iron formulations based on S-Chelate-O technology produce nutrient solutions that remain relatively transparent when dissolved, allowing ultraviolet treatment systems to function effectively.
These characteristics allow the nutrient solution to pass through UV systems without interfering with water treatment.
For greenhouse growers operating recirculating irrigation systems, compatibility between fertilisers and sterilisation technologies is essential.
Iron Nutrition for Large Scale Greenhouse Fertigation Systems
Greenhouse horticulture has evolved rapidly over the past two decades. Many modern facilities operate on a scale that rivals industrial production systems.
These facilities rely on precise environmental control together with automated fertigation systems.
Nutritional Requirements of Modern Greenhouses
Large greenhouse complexes require fertilisers that deliver consistent performance across many hectares of crop production.
Key requirements include:
- High solubility
- Reliable nutrient stability
- Compatibility with irrigation equipment
- Minimal sediment formation
- Compatibility with water sterilisation systems
S-Chelate-O Technology in Global Production
S-Chelate-O Iron technology is used in micronutrient formulations designed for greenhouse production systems around the world.
The technology supports growers who need micronutrient solutions that integrate seamlessly into complex irrigation networks.
By combining solubility, stability and UV compatibility, S-Chelate-O Iron helps support efficient nutrient delivery within modern greenhouse environments.
Conclusion: Smarter Iron Nutrition for Modern Horticulture
Iron remains one of the most important micronutrients for plant health and crop productivity. In greenhouse production systems, maintaining consistent iron availability is essential for preventing chlorosis and supporting healthy plant development.
Traditional iron fertilisers can struggle to meet the demands of modern fertigation systems. Issues such as poor solubility, precipitation and incompatibility with irrigation technologies can limit their effectiveness.
Chelated micronutrients provide a more reliable solution. Through advanced chelation technology, S-Chelate Plant Nutrition has developed S-Chelate-O Iron to maintain stable and biologically available iron across a wide range of growing conditions.
S-Chelate-O Iron provides growers with a micronutrient solution designed for modern irrigation systems where solubility, stability and compatibility with water treatment technologies are essential.
As greenhouse agriculture continues to expand globally, nutrient solutions that combine solubility, stability and compatibility with advanced irrigation technologies will play an increasingly important role in sustainable crop production.
Discover how S-Chelate Plant Nutrition supports greenhouse growers with advanced chelation technology for reliable micronutrient delivery. Buy Now.

